|
|
|
|
|
菌群移植治疗改善自闭症谱系障碍与胃肠功能紊乱 | Microbiome |
|
|
论文标题:Microbiota Transfer Therapy alters gut ecosystem and improves gastrointestinal and autism symptoms: an open-label study
期刊:Microbiome
作者:Dae-Wook Kang†, James B. Adams†, Ann C. Gregory†, Thomas Borody, Lauren Chittick, Alessio Fasano, Alexander Khoruts, Elizabeth Geis, Juan Maldonado, Sharon McDonough-Means, Elena L. Pollard, Simon Roux, Michael J. Sadowsky, Karen Schwarzberg Lipson, Matthew B. SullivanE, J. Gregory Caporaso and Rosa Krajmalnik-Brown
发表时间:2017/01/23
数字识别码:10.1186/s40168-016-0225-7
原文链接:https://microbiomejournal.biomedcentral.com/articles/10.1186/s40168-016-0225-7?utm_source=other&utm_medium=other&utm_content=null&utm_campaign=BSCN_2_DD_Article_Scinet
微信链接:https://mp.weixin.qq.com/s/h6Z6wbNffsUoD8gaA3WXxA
自闭症谱系障碍(ASD)是一种复杂的神经生物学疾病,影响患者的社会交往,导致其行为、兴趣或活动受限,呈现重复、刻板的行为模式。ASD的发病原因目前仍未被充分阐明。然而,ASD患儿常常表现出胃肠道症状,并且这种症状与ASD的严重程度相关,这表明人类肠道中数以百亿计的细菌(即肠道菌群)很有可能与ASD有关联。已有几项研究显示ASD患儿的肠道菌群异常。已在ASD小鼠模型中验证了肠道微生物组与ASD的关联,研究发现肠道微生物组与小鼠的异常代谢产物或行为存在联系。一项针对ASD患儿的研究也发现,口服不可吸收抗生素进行治疗能够短暂地改善ASD患儿的胃肠道症状和ASD症状。
在发表于Microbiome的一篇文章Microbiota Transfer Therapy alters gut ecosystem and improves gastrointestinal and autism symptoms: an open-label study中,由美国亚利桑那州立大学Dae-Wook Kang等组成的研究团队开展了一项小规模的双盲临床试验,在18名ASD患儿中评估了菌群移植治疗(MTT)对肠道菌群构成以及胃肠道症状和ASD症状的影响。

图一:研究设计时间线
菌群移植治疗包括2周的抗生素治疗,1次肠道清洗,随后再进行粪便微生物移植(起始高剂量,随后逐日降低剂量,持续7-8周)。胃肠症状评定量表评估显示,治疗结束时,患儿的便秘、腹泻、消化不良与腹痛等胃肠道症状减少了约80%,并且这些改善在治疗后可持续8周。同样,临床评估显示,治疗后患儿的ASD症状显著改善,且在治疗结束后8周改善仍可持续。菌群与噬菌体深度测序分析显示,供体微生物群成功地部分定植,患儿的肠道菌群环境也得到改善。具体而言,随访8周期间,患儿总体的肠道菌群多样性以及双歧杆菌属、普雷沃菌属、脱硫弧菌属的丰度均有显著提升。
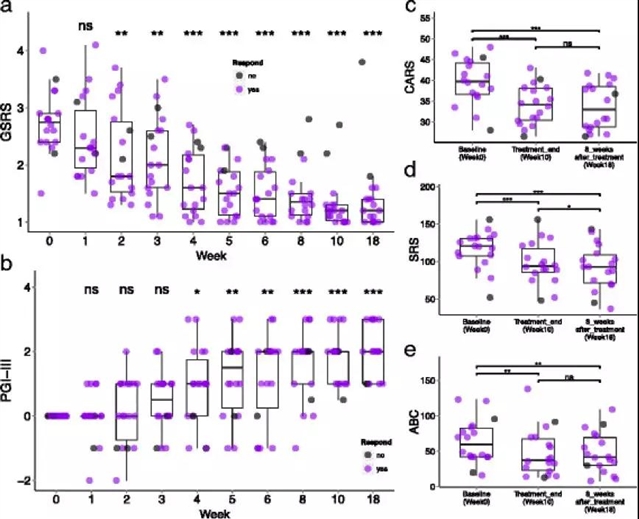
图二:18个ASD患儿与胃肠道和ASD相关的症状
这种探索性、持续时间较长的治疗方案似乎是一种很有前景的治疗方法,可改变ASD患儿的肠道微生物组与病毒组,从而改善其胃肠道症状与ASD行为表现,且胃肠道症状、ASD症状以及肠道微生物群的改善在治疗结束后至少持续8周,这表明该治疗方案具有长期效应。
摘要:
Background
Autism spectrum disorders (ASD) are complex neurobiological disorders that impair social interactions and communication and lead to restricted, repetitive, and stereotyped patterns of behavior, interests, and activities. The causes of these disorders remain poorly understood, but gut microbiota, the 1013 bacteria in the human intestines, have been implicated because children with ASD often suffer gastrointestinal (GI) problems that correlate with ASD severity. Several previous studies have reported abnormal gut bacteria in children with ASD. The gut microbiome-ASD connection has been tested in a mouse model of ASD, where the microbiome was mechanistically linked to abnormal metabolites and behavior. Similarly, a study of children with ASD found that oral non-absorbable antibiotic treatment improved GI and ASD symptoms, albeit temporarily. Here, a small open-label clinical trial evaluated the impact of Microbiota Transfer Therapy (MTT) on gut microbiota composition and GI and ASD symptoms of 18 ASD-diagnosed children.
Results
MTT involved a 2-week antibiotic treatment, a bowel cleanse, and then an extended fecal microbiota transplant (FMT) using a high initial dose followed by daily and lower maintenance doses for 7–8 weeks. The Gastrointestinal Symptom Rating Scale revealed an approximately 80% reduction of GI symptoms at the end of treatment, including significant improvements in symptoms of constipation, diarrhea, indigestion, and abdominal pain. Improvements persisted 8 weeks after treatment. Similarly, clinical assessments showed that behavioral ASD symptoms improved significantly and remained improved 8 weeks after treatment ended. Bacterial and phagedeep sequencing analyses revealed successful partial engraftment of donor microbiota and beneficial changes in the gut environment. Specifically, overall bacterial diversity and the abundance of Bifidobacterium, Prevotella, and Desulfovibrio among other taxa increased following MTT, and these changes persisted after treatment stopped (followed for 8 weeks).
Conclusions
This exploratory, extended-duration treatment protocol thus appears to be a promising approach to alter the gut microbiome and virome and improve GI and behavioral symptoms of ASD. Improvements in GI symptoms, ASD symptoms, and the microbiome all persisted for at least 8 weeks after treatment ended, suggesting a long-term impact.
Trial registration
This trial was registered on the ClinicalTrials.gov, with the registration number NCT02504554
阅读论文全文请访问:
https://microbiomejournal.biomedcentral.com/articles/10.1186/s40168-016-0225-7?utm_source=other&utm_medium=other&utm_content=null&utm_campaign=BSCN_2_DD_Article_Scinet
期刊介绍:
The central purpose of Microbiome(https://microbiomejournal.biomedcentral.com/) is to unite investigators conducting microbiome research in environmental, agricultural, and biomedical arenas.
(来源 :科学网)
特别声明:本文转载仅仅是出于传播信息的需要,并不意味着代表本网站观点或证实其内容的真实性;如其他媒体、网站或个人从本网站转载使用,须保留本网站注明的“来源”,并自负版权等法律责任;作者如果不希望被转载或者联系转载稿费等事宜,请与我们接洽。