近日,美国加州大学Jingjing Li及其小组研制了人类母胎界面的单细胞时空解剖。该研究于2026年4月8日发表于国际一流学术期刊《自然》杂志上。
在这里,该课题组人员通过整合大规模配对单核转录组和染色质可及性分析与亚微米分辨率空间转录组学和CODEX多重蛋白成像,生成了从妊娠早期到足月正常妊娠期间的人类母胎界面的全面图谱,大大提高了先前研究的时空分辨率。该框架描述了常见的和短暂的细胞类型、状态和空间壁龛,重构了引导细胞滋养细胞和蜕质细胞分化的转录程序,并解决了构建该界面的循环结构单元。
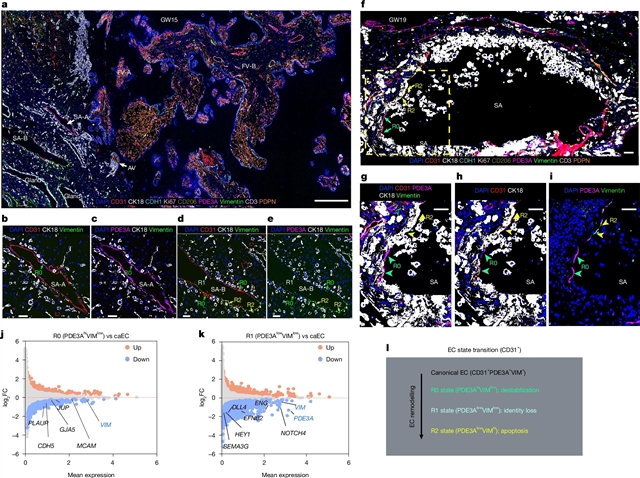
课题组研究人员在细胞滋养层介导的螺旋动脉重塑过程中发现了以前未被识别的动脉内皮状态转变,并开发了一种机器学习模型,通过转录组特征预测细胞滋养层的侵袭性。小组进一步发现了一种通过内源性大麻素信号在人母胎界面抑制细胞滋养细胞侵袭的蜕质细胞亚型。通过整合图谱与全基因组关联数据,课题组研究人员确定了母体和胎儿细胞最易患先兆子痫、早产或流产。该研究为人类胎盘和蜕膜的单细胞多组学提供了全面的空间分辨参考,并为解码其正常和紊乱的发育提供了框架。
据悉,人类母胎界面的特征是母胎细胞和胎儿细胞的嵌合。然而,潜在的细胞、分子和空间程序仍未完全确定。
附:英文原文
Title: Single-cell spatiotemporal dissection of the human maternal–fetal interface
Author: Wang, Cheng, Zhou, Yan, Wang, Yuejun, Guha, Tuhin Kumar, Luo, Zhida, Mustafaraj, Anxhela, McIntyre, Tara I., Schwab, Marisa E., Davidson, Brittany R., Reeder, Gabriella C., Wong, Ronald J., England, Sarah K., Gonzalez, Juan M., Blelloch, Robert, Combes, Alexis J., Giudice, Linda C., Erlebacher, Adrian, MacKenzie, Tippi C., Stevenson, David K., Shaw, Gary M., Snyder, Michael P., Sun, Xiaofei, Winn, Virginia D., Fisher, Susan J., Li, Jingjing
Issue&Volume: 2026-04-08
Abstract: The human maternal–fetal interface is characterized by mosaic intermingling of maternal and fetal cells1. Yet the underlying cellular, molecular and spatial programmes remain incompletely defined. Here we generate a comprehensive atlas of the human maternal–fetal interface across normal pregnancies from early gestation to term by integrating large-scale paired single-nucleus transcriptomic and chromatin accessibility profiling with submicrometre-resolution spatial transcriptomics and CODEX multiplex protein imaging2, substantially boosting the spatiotemporal resolution of prior research3. This framework delineates common and transient cell types, states and spatial niches across the fetal and maternal compartments, reconstructs transcriptional programmes that guide cytotrophoblast and decidual stromal cell differentiation, and resolves recurrent architecture structural units that build this interface. We identify previously unrecognized arterial endothelial state transitions during cytotrophoblast-mediated spiral artery remodelling and develop a machine learning model that predicts cytotrophoblast invasiveness from transcriptomic signatures. We further discover a decidual stromal cell subtype that suppresses cytotrophoblast invasion via endocannabinoid signalling at the human maternal–fetal interface. By integrating the atlas with genome-wide association data, we pinpoint maternal and fetal cells that are most vulnerable to pre-eclampsia, preterm birth or miscarriage. This resource provides a comprehensive spatially resolved single-cell multiomic reference of the human placenta and decidua and offers a framework for decoding their normal and disordered development.
DOI: 10.1038/s41586-026-10316-x
Source: https://www.nature.com/articles/s41586-026-10316-x
Nature:《自然》,创刊于1869年。隶属于施普林格·自然出版集团,最新IF:69.504
官方网址:http://www.nature.com/
投稿链接:http://www.nature.com/authors/submit_manuscript.html
