近日,北京大学李厚华团队报道了利用仿生骨架编辑策略的短肽的对映选择性全合成。2026年4月7日出版的《德国应用化学》杂志发表了这项成果。
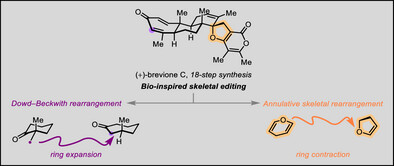
受螺环二萜吡喃酮生物合成中骨架编辑转化的启发,研究组实现了海洋萜类化合物breviones B、C和N的对映选择性全合成。该合成策略包含一系列仿生骨架编辑转化,其中包括构建七元A环的Dowd–Beckwith扩环反应,以及将萜烯与α-吡喃酮片段偶联成螺环二萜吡喃酮骨架的环化骨架重排。
后一种转化涉及一种新颖的二氢吡喃至二氢呋喃的环收缩策略,该策略已被证明适用于多种萜烯和吡喃酮片段。此外,在合成过程中,研究组还开发了一种SnCl4介导的烯基β-酮酯分子内氢烷基化反应,能够同时构建稠合的B/C环系并形成三个连续的手性中心。
附:英文原文
Title: Enantioselective Total Synthesis of Breviones Utilizing a Bio-Inspired Skeletal Editing Strategy
Author: Yaqian Liu, Yuanjun Zhou, Yaoyao Xu, Shuangning Liu, Qingyang Huang, Hongli Jia, Houhua Li
Issue&Volume: 2026-04-07
Abstract: Inspired by skeletal editing transformations in the biosynthesis of spiro diterpene pyrones, we herein present an enantioselective total synthesis of the meroditerpenoids breviones B, C, and N. Our synthetic strategy features a series of bio-inspired skeletal editing transformations, including a Dowd–Beckwith ring expansion to construct the seven-membered A-ring, and an annulative skeletal rearrangement that couples terpene and α-pyrone motifs into the spiro diterpene pyrone framework. This latter transformation involves a novel dihydropyran-to-dihydrofuran ring contraction, a strategy that has proven applicable to a diverse range of terpene and pyrone fragments. In addition, during the synthesis, we also developed a SnCl4-mediated intramolecular hydroalkylation of alkenyl β-keto esters, which enables the concurrent assembly of the fused B/C ring system along with three contiguous stereocenters.
DOI: 10.1002/anie.4698250
Source: https://onlinelibrary.wiley.com/doi/10.1002/anie.4698250
Angewandte Chemie:《德国应用化学》,创刊于1887年。隶属于德国化学会,最新IF:16.823
官方网址:https://onlinelibrary.wiley.com/journal/15213773
投稿链接:https://www.editorialmanager.com/anie/default.aspx
