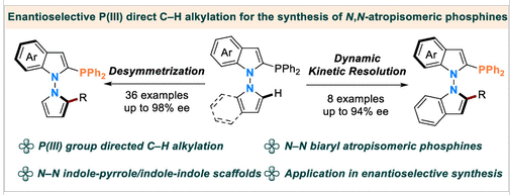
近日,青岛大学刘人荣团队报道了对映选择性铱催化C-H键烷基化合成N-N阿托普二聚体膦。该项研究成果发表在2026年4月7日出版的《美国化学会志》上。
在过去的几十年中,阻旋异构的联芳基膦已成为手性配体、生物活性分子及药物候选物设计中的优势骨架。然而,尽管潜力巨大,N-N阻旋异构膦的对映选择性合成仍发展不足。
研究组报道了一种铱催化的对映选择性C-H烷基化反应,以膦(III)基团作为导向基团,用于构建N-N阻旋异构的单膦配体。该方法能够便捷地获得多种N-N连接的吲哚-吡咯及吲哚-吲哚骨架结构,对映选择性高达98% ee。此外,通过一系列合成转化和催化应用,验证了该方法的实用性。
附:英文原文
Title: Enantioselective Iridium-Catalyzed C–H Bond Alkylation for the Synthesis of N–N Atropisomeric Phosphines
Author: Si-Hao Fu, Rui-Jing Pang, Yi Yue, Long-Long Xi, Jia Feng, Ren-Rong Liu
Issue&Volume: April 7, 2026
Abstract: Over the past few decades, atropisomeric biaryl phosphines have emerged as privileged scaffolds in the design of chiral ligands, bioactive molecules, and drug candidates. However, despite their considerable potential, the enantioselective synthesis of N–N atropisomeric phosphines remains underdeveloped. Herein, we report an iridium-catalyzed, enantioselective C–H alkylation for constructing N–N atropisomeric monophosphines, employing a phosphine(III) group as a directing motif. This method enables ready access to a diverse range of N–N-linked indole–pyrrole and indole–indole frameworks with up to 98% ee. Furthermore, the utility of this approach is demonstrated through a series of synthetic transformations and catalytic applications.
DOI: 10.1021/jacs.5c22411
Source: https://pubs.acs.org/doi/full/10.1021/jacs.5c22411
JACS:《美国化学会志》,创刊于1879年。隶属于美国化学会,最新IF:16.383
官方网址:https://pubs.acs.org/journal/jacsat
投稿链接:https://acsparagonplus.acs.org/psweb/loginForm?code=1000
