近日,瑞士苏黎世大学Deliz Liang, Alexandria团队研究了铜金属酶中非规范氨基酸的疏水调谐。2026年4月13日,《自然-化学》杂志发表了这一成果。
疏水性控制着蛋白质和酶功能的诸多方面。尽管使用经典氨基酸可以在一定程度上实现疏水调控,但引入非天然氨基酸能进一步扩展这种能力,从而赋予新的或改进的功能。
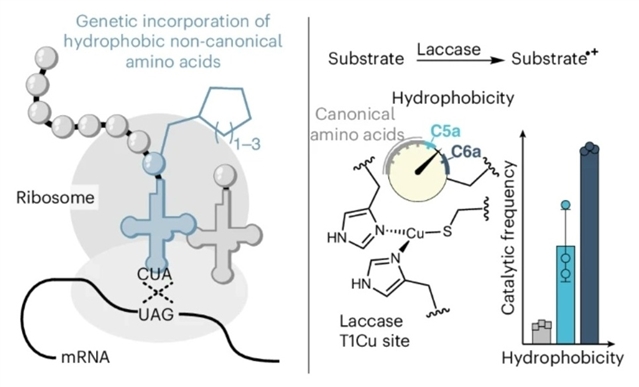
研究组设计了一对氨酰-tRNA合成酶/tRNA体系,用于对一组体积庞大、疏水性强的氨基酸(即环戊基丙氨酸、环己基丙氨酸和环庚基丙氨酸)进行位点特异性遗传编码。作为概念验证,研究组展示了基于非天然氨基酸的疏水调控在工程化细菌漆酶中的实用性——漆酶既是一种典型的金属酶,也是工业过程中具有高价值的催化剂。
所获得的突变显著提升了催化活性,尤其是转换频率和总转换数。为了理解这种功能改进,研究组检测了氧化还原电位、电子光谱以及构效关系。将传统的定向进化与非天然氨基酸工程化相结合,进一步提升了催化性能,并通过分析这两种方法所带来的变化,对此进行了情境化阐释。
附:英文原文
Title: Hydrophobic tuning with non-canonical amino acids in a copper metalloenzyme
Author: Fischer, Sandro, Natter Perdiguero, Anton, Lau, Kelvin, Deliz Liang, Alexandria
Issue&Volume: 2026-04-13
Abstract: Hydrophobicity controls many aspects of protein and enzyme function. Although hydrophobic tuning can be somewhat achieved with canonical amino acids, the incorporation of non-canonical amino acids further extends this ability to enable new and improved functionality. Here we engineer an aminoacyl-tRNA synthetase/tRNA pair for the site-specific genetic encoding of a set of bulky, hydrophobic amino acids, namely cyclopentylalanine, cyclohexylalanine and cycloheptylalanine. As a proof of concept, we demonstrate the utility of hydrophobic tuning based on non-canonical amino acids (ncAAs) to engineer a bacterial laccase, which is both a classical metalloenzyme and a high-value catalyst for industrial processes. The resulting mutations substantially improved the catalytic activity, particularly the turnover frequency and total turnover number. To understand this improved functionality, the redox potentials, electronic spectra and structure–function relationships were examined. Combining traditional directed evolution with ncAA-based engineering resulted in further improvements in catalysis, which were contextualized by analysing the changes imparted from these two methods.
DOI: 10.1038/s41557-026-02116-7
Source: https://www.nature.com/articles/s41557-026-02116-7
Nature Chemistry:《自然—化学》,创刊于2009年。隶属于施普林格·自然出版集团,最新IF:24.274
官方网址:https://www.nature.com/nchem/
投稿链接:https://mts-nchem.nature.com/cgi-bin/main.plex
