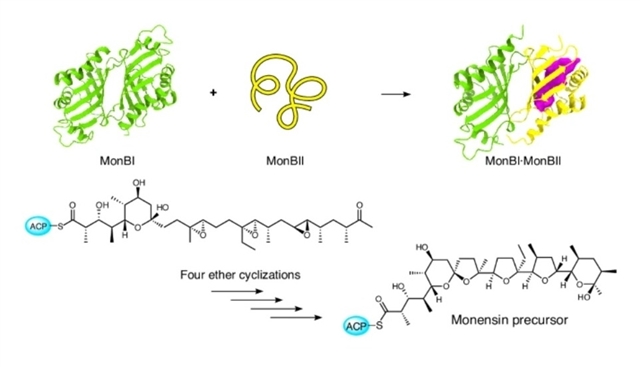
近日,日本北海道大学Toyoyuki Ose团队报道了成对的聚醚环氧水解酶系统赋予了一种可塑酶以催化连续环化级联反应的能力。2026年4月16日,《自然-化学》杂志发表了这一成果。
离子载体聚醚是一类主要的聚酮来源天然产物,其分子骨架特征在于由四氢呋喃和四氢吡喃环阵列构成。然而,其生物合成的确切机制——据推测是通过连续的环氧开环与醚环化级联反应生成两个以上的醚环——仍不明确。
研究组探究了莫能菌素的生物合成,并揭示了多聚醚环氧水解酶异源二聚体复合物(MonBI·MonBII)的不可或缺性,其中MonBII提供唯一的活性位点。结构分析表明,MonBII具有一个异常庞大的空腔,能够使含有γ-羟基三环氧部分底物发生连续环化。
该空腔的形成归因于MonBII显著的柔性:它仅在MonBI存在下,从主要以无序状态转变为活性折叠构象,从而实现剧烈的结构转变。鉴于MonBI和MonBII型酶在进化上的高度保守性,该研究强调了一种统一的配对机制,即其中一个亚基充当分子模具,促进其伴侣蛋白的折叠过程并稳定其结构以行使催化功能。
附:英文原文
Title: A system of paired polyether epoxide hydrolases enables a mouldable enzyme for consecutive ring cyclization cascades
Author: Yabuno, Nana, Minami, Atsushi, Ozaki, Taro, Owada, Yuki, Sawada, Kohei, Arai, Ayaka, Sato, Shusuke, Sugiyama, Aoi, Tadokoro, Takashi, Aizawa, Tomoyasu, Dosen, Takuya, Nomai, Tomo, Matsumaru, Takanori, Liu, Junyang, Ye, Tao, Kodama, Atsuji, Uchiyama, Susumu, Hengphasatporn, Kowit, Shigeta, Yasuteru, Saio, Tomohide, Maenaka, Katsumi, Yao, Min, Kumeta, Hiroyuki, Oikawa, Hideaki, Ose, Toyoyuki
Issue&Volume: 2026-04-16
Abstract: Ionophore polyethers, a major class of polyketide-derived natural products, are characterized by molecular skeletons featuring arrays of tetrahydrofuran and tetrahydropyran rings. However, the precise mechanism underlying their biosynthesis, suggested to occur by a sequential epoxide-opening and ether cyclization cascade to generate more than two ether rings, remains elusive. Here we explore the biosynthesis of monensin and reveal the indispensability of a heterodimeric assembly of polyether epoxide hydrolases (MonBI·MonBII), with MonBII providing the sole active site. Structural analysis demonstrated that MonBII harbours an unusually large cavity, enabling the consecutive cyclization of substrates containing γ-hydroxy triepoxide moieties. This cavity is formed by the remarkable flexibility of MonBII, which undergoes a dramatic structural transition from a predominantly disordered state to its active folded conformation exclusively in the presence of MonBI. Given the widespread conservation of MonBI- and MonBII-type enzymes, this study emphasizes a unified pairing mechanism, wherein one protomer functions as a molecular mould, facilitating the folding process and stabilizing the structure of its partner for catalysis.
DOI: 10.1038/s41557-026-02122-9
Source: https://www.nature.com/articles/s41557-026-02122-9
Nature Chemistry:《自然—化学》,创刊于2009年。隶属于施普林格·自然出版集团,最新IF:24.274
官方网址:https://www.nature.com/nchem/
投稿链接:https://mts-nchem.nature.com/cgi-bin/main.plex
