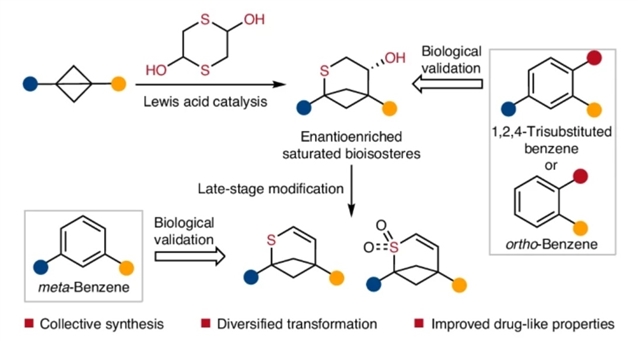
近日,湖南大学冯见军团队报道了以双丁烷为原料合成1,2,4-三取代、间取代和邻取代芳烃生物异构体。相关论文发表在2026年3月3日出版的《自然-化学》杂志上。
在药物化学领域,用富含C(sp3)结构的生物电子等排体替代苯环以改善化合物的类药性,是一项颇具吸引力的策略。尽管许多笼状烃已被验证为邻位和间位二取代苯的有效生物电子等排体,但对于药物中第二常见的苯环结构类型——1,2,4-三取代苯的三维类似物,由于向量保真度和对映选择性获取仍是艰巨挑战,其开发进展缓慢。
研究组报道了一条合成(对映体纯)2-硫代双环[3.1.1]庚烷的实用路线,该合成通过双环[1.1.0]丁烷与1,4-二噻烷-2,5-二醇的环加成反应实现。此方法生成的环加成产物具有两个和三个伸出向量,分别可作为邻位取代苯和1,2,4-三取代苯的理想生物电子等排体。此外,这些环加成产物可进一步转化为多样化的化学空间,包括1,5-二取代硫代双环[3.1.1]庚烯。
晶体学分析、药代动力学性质比较,以及二氟尼柳、水杨酰苯胺和抗癌药索尼吉布与其三维thia-BCHep类似物的生物活性评估表明,所获得的thia-BCHep类化合物可为药物发现项目中的1,2,4-三取代、间位和邻位取代苯环提供新型替代基团。
附:英文原文
Title: Collective synthesis of 1,2,4-trisubstituted, meta- and ortho-substituted arene bioisosteres from bicyclobutanes
Author: Wu, Feng, Wang, Ji-Jie, Xiao, Yuanjiu, Peng, Quanxin, Li, Yu-Jie, Peng, Keren, Han, Qianlan, Wei, Mengran, Qian, Yu, Zhang, Wei, Wang, Guoqiang, Feng, Jian-Jun
Issue&Volume: 2026-03-03
Abstract: Replacing benzene rings with C(sp3)-rich bioisosteres to yield compounds with improved drug-like properties is an attractive strategy in medicinal chemistry. While many caged hydrocarbons have been validated as bioisosteres of ortho- and meta-disubstituted benzenes, 3D analogues of 1,2,4-trisubstituted benzenes—the second most prevalent benzenoid pattern in drugs—remain elusive because vector fidelity and enantioselective access are still formidable challenges. Here we report a practical route to (enantiomerically pure) 2-thiabicyclo[3.1.1]heptanes (thia-BCHeps) by cycloadditions of bicyclo[1.1.0]butanes with 1,4-dithiane-2,5-diol. This method produces cycloadducts with two and three exit vectors, which serve as promising bioisosteres for ortho-substituted and 1,2,4-trisubstituted benzenes, respectively. Moreover, the cycloadducts can be transformed into a diverse chemical space, including 1,5-disubstituted thiabicyclo[3.1.1]heptenes. Crystallographic analysis and a comparison of the pharmacokinetic properties, along with an evaluation of the biological activity of diflunisal, salicylanilide and the anticancer drug sonidegib, in relation to their 3D thia-BCHep analogues, demonstrate that the thia-BCHeps obtained can provide new surrogates for 1,2,4-trisubstituted, meta- and ortho-substituted benzene rings in drug discovery programmes.
DOI: 10.1038/s41557-026-02097-7
Source: https://www.nature.com/articles/s41557-026-02097-7
Nature Chemistry:《自然—化学》,创刊于2009年。隶属于施普林格·自然出版集团,最新IF:24.274
官方网址:https://www.nature.com/nchem/
投稿链接:https://mts-nchem.nature.com/cgi-bin/main.plex
