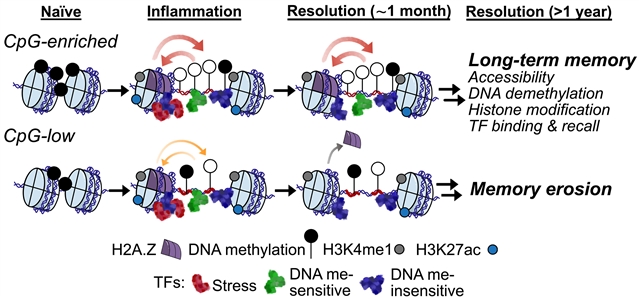
该课题组研究人员表明,在小鼠中,表皮干细胞储存了牛皮癣样皮肤耀斑的终身功能表观遗传记录。应用深度学习来研究这些染色质动力学,研究小组发现CpG二核苷酸密度是记忆持久性的主要驱动因素。尽管炎症诱导的转录因子打开和建立记忆是不必要的,但CpG富集序列随后成为必不可少的,通过整合DNA去甲基化、甲基化敏感转录因子、序列固有核小体去亲和力和核小体不稳定组蛋白变体H2A.Z,加强了细胞世代的可及性。它们一旦被炎症诱导的转录因子激活,DNA序列就会协调持久的平衡,将持久的记忆传递给压力敏感基因,并在回忆时深刻影响组织的适应性。
据悉,组织储存着炎症的记忆,这提高了对未来各种攻击的敏感性。这些适应性是否以及如何通过时间和细胞分裂得以维持,人们仍然知之甚少。
附:英文原文
Title: Distinctive DNA sequence features define epigenetic longevity of inflammatory memory
Author: Christopher J. Cowley, Sairaj M. Sajjath, Luis F. Soto-Ugaldi, Mara Steiger, Samantha B. Larsen, Thomas Carroll, Douglas Barrows, Alexandra Mattei, Kevin A. U. Gonzales, Wei Wang, Kevin Li, Alexander Meissner, Helene Kretzmer, Dana Pe’er, Elaine Fuchs
Issue&Volume: 2026-03-26
Abstract: Tissues harbor memories of inflammation, which heighten sensitivity to diverse future assaults. Whether and how these adaptations are sustained through time and cell division remain poorly understood. We show that in mice, epidermal stem cells store lifelong, functional epigenetic records of psoriasis-like skin flares. Applying deep learning to investigate these chromatin dynamics, we unearth CpG dinucleotide density as a major driver of memory persistence. Although unnecessary for inflammation-induced transcription factors to open and establish memories, CpG-enriched sequences thereafter become essential, reinforcing accessibility across cellular generations by integrating DNA demethylation, methylation-sensitive transcription factors, sequence-intrinsic nucleosome disaffinity, and the nucleosome-destabilizing histone variant H2A.Z. Thus, once activated by inflammation-induced transcription factors, DNA sequences orchestrate persistent poise, imparting long-lasting memory to stress-sensitive genes and profoundly affecting tissue fitness upon recall.
DOI: adz6830
Source: https://www.science.org/doi/10.1126/science.adz6830
