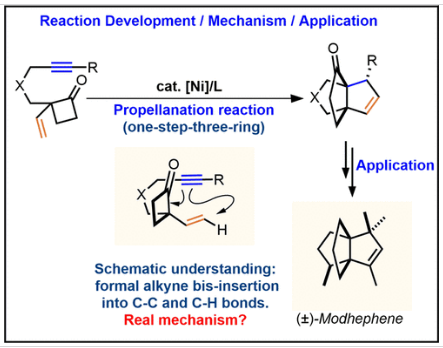
近日,北京大学余志祥团队报道了镍催化乙烯基环丁酮的一步三环推进反应:反应发展、机理和莫庚烯的形式化合成。该项研究成果发表在2026年3月24日出版的《美国化学会志》上。
[3.3.3]螺桨烷骨架存在于多种天然产物中,是合成化学中备受关注的三维结构。传统上,这类化合物通过逐环构筑策略合成,该策略不仅步骤冗长,而且构建目标结构中相邻的桥头季碳中心极具挑战性。
研究组报道了一种镍催化的一步三环螺桨烷化反应,可从线性炔基乙烯基环丁酮直接构筑[3.3.3]螺桨烷核心骨架。该方法采用Ni(COD)2与P(Ad)3为催化体系,具有广泛的底物适用性。研究组报道了一种镍催化的一步三环推进反应,该反应由直链炔乙烯基环丁酮构建了[3.3.3]推进烷核。该螺桨烷化反应可视为炔烃对环丁酮中C–C键及底物乙烯基中C–H键的双卡宾插入过程,但实际反应路径经DFT计算与氘代标记实验证实为:炔烃与羰基的氧化环金属化、Cope重排、β-氢消除、三烯醇环化及还原消除。
同时,通过理论计算研究了从Ni(COD)2到膦配体与底物配位的镍配合物的详细配体交换过程(反应引发过程)。研究组还分析了底物中连接基团对螺桨烷化反应及乙烯基环丁酮重排为环己烯酮副反应的影响,发现NTs、O等吸电子连接基团有利于螺桨烷化反应,而与NTs相比吸电子能力较弱的CH2、NBn等中性连接基团则会降低这种选择性。
附:英文原文
Title: One-Step-Three-Ring Propellanation Reaction of Yne-Vinylcyclobutanones Catalyzed by Nickel: Reaction Development, Mechanism, and Formal Synthesis of Modhephene
Author: Jiguo Ma, Bing-Wen Li, Zhi-Xiang Yu
Issue&Volume: March 24, 2026
Abstract: The [3.3.3]propellane scaffold, present in various natural products, is a three-dimensional structure of interest in synthetic chemistry. Traditionally, these compounds are synthesized through a ring-by-ring strategy that is tedious (long steps of synthesis) and challenging (building two adjacent bridgehead quaternary centers of the target motifs is a formidable task). Herein, we report a nickel-catalyzed one-step, three-ring propellanation reaction that constructs the [3.3.3] propellane core from linear yne-vinylcyclobutanones. This method employs Ni(COD)2 and P(Ad)3 as the catalytic system and exhibits a broad substrate scope. The utility of this reaction has been further demonstrated through the formal synthesis of modhephene, a natural product featuring a [3.3.3] propellane skeleton. This propellanation reaction can be envisioned as biscarbene insertion of alkyne into the C–C bond in cyclobutanone and the C–H of the vinyl group in the substrates, but it actually takes place through oxidative cyclometalation of the alkyne and carbonyl group, Cope rearrangement, β-hydrogen elimination, trienolate cyclization, and reductive elimination, supported by DFT calculations and a deuterium labeling experiment. The detailed ligand exchange reaction (reaction initiation process) from Ni(COD)2 to the Ni complex coordinated by the phosphine ligand and substrate, has also been studied computationally. How the tether group in the substrates affects the propellanation reaction and the side rearrangement reaction of vinylcyclobutanones to cyclohexenones has been analyzed, finding that electron-withdrawing tethers such as NTs and O favor the propellanation reaction, while the electron-neutral tether such as CH2 and NBn tether, which is less electron-withdrawing compared to NTs, reduce this preference.
DOI: 10.1021/jacs.5c21425
Source: https://pubs.acs.org/doi/abs/10.1021/jacs.5c21425
JACS:《美国化学会志》,创刊于1879年。隶属于美国化学会,最新IF:16.383
官方网址:https://pubs.acs.org/journal/jacsat
投稿链接:https://acsparagonplus.acs.org/psweb/loginForm?code=1000
