近日,西湖大学叶宇轩团队报道了烯还原酶转化成异构酶助力对映发散合成异丙酸盐。相关论文于2026年3月19日发表在《美国化学会杂志》上。
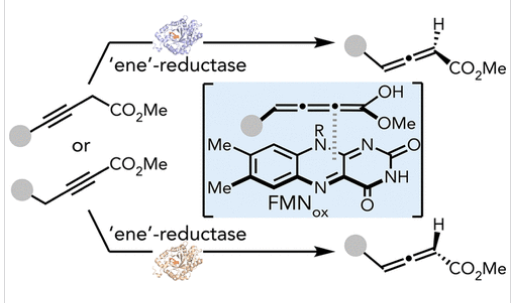
通过质子转移实现非手性炔酸酯的酶促不对称异构化来合成手性联烯酸酯,是一项极具吸引力却尚未实现的转化,其主要障碍在于天然异构酶存在显著产物抑制效应。为突破这一瓶颈,研究组将传统上属于氧化还原酶家族的黄素依赖性"烯"还原酶(EREDs)改造为氧化还原中性的立体选择性1,3-质子转移酶。
通过定向进化技术,来自嗜高温红藻(Galdieria sulphuraria)的ERED(GsOYE)经改造后获得了一系列新型异构酶,可高效催化3-丁炔酸酯与2-丁炔酸酯的异构化反应,以优异的对映发散性合成多种手性联烯酸酯,且产物抑制效应可忽略不计。
所合成的联烯酸酯进一步应用于手性传递的[2+2]、[3+2]及[4+2]环加成反应,成功构建了含多达三个立体中心的复杂多环化合物。机制研究表明,定向进化使GsOYE突变体能够通过单碱基或双碱基机制催化反应,这种功能分化在小分子催化剂中极为罕见。该研究拓展了EREDs的催化功能边界,为通过质子转移催化构建轴手性化合物建立了新的生物催化平台。
附:英文原文
Title: Repurposing “Ene”-Reductase to Isomerase for Enantiodivergent Synthesis of Allenoates
Author: Heli Cheng, Kun Zhang, Pen Chang, Tianyu Zhu, Yuxuan Ye
Issue&Volume: March 19, 2026
Abstract: The synthesis of chiral allenoates through enzymatic asymmetric isomerization of achiral alkynoates via proton transfer is a highly desirable yet unachieved transformation, primarily due to significant product inhibition encountered with natural isomerases. To circumvent this, we repurposed flavin-dependent “ene”-reductases (EREDs), traditionally oxidoreductases, for redox-neutral stereoselective 1,3-proton transfer. Directed evolution of an ERED from Galdieria sulphuraria (GsOYE) generated a panel of new-to-nature isomerases proficient in isomerizing both 3-butynoates and 2-butynoates, affording diverse chiral allenoates with excellent enantiodivergence and negligible product inhibition. The resulting allenoates were leveraged in chirality transfer [2 + 2], [3 + 2], and [4 + 2] cycloadditions to create complex polycycles with up to three stereogenic centers. Mechanistic studies revealed that directed evolution yielded GsOYE mutants capable of operating via either one- or two-base mechanisms, a functional divergence rarely seen with small-molecule catalysts. This study expanded the catalytic repertoire of EREDs, establishing a biocatalytic platform for proton transfer catalysis to set axial chirality.
DOI: 10.1021/jacs.5c19848
Source: https://pubs.acs.org/doi/abs/10.1021/jacs.5c19848
JACS:《美国化学会志》,创刊于1879年。隶属于美国化学会,最新IF:16.383
官方网址:https://pubs.acs.org/journal/jacsat
投稿链接:https://acsparagonplus.acs.org/psweb/loginForm?code=1000
