近日,济南大学王斌团队报道了Rieske双加氧酶的功能模拟实现醌的催化不对称顺式二羟基化。2026年3月18日出版的《美国化学会志》发表了这项成果。
开发基于地球丰富金属催化剂的高效实用催化不对称顺式双羟基化反应仍是一项重大挑战;1,4-醌的顺式双羟基化反应是获得手性顺式-1,2-二醇的关键途径,这类化合物是生物活性天然产物和药物的重要骨架。
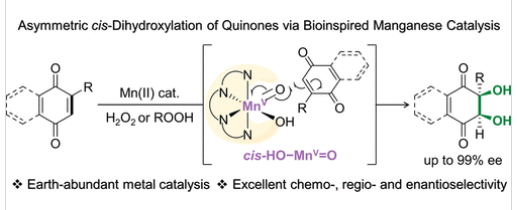
受Rieske双加氧酶催化芳基和烯基C=C键不对称顺式双羟基化反应的启发,研究组提出了一种模拟生物策略,用于实现1,4-醌的不对称顺式双羟基化反应。研究组报道了一种廉价易得的非血红素锰配合物,其配体为含有刚性手性二胺骨架和大位阻三异丙基硅基的四齿氨基吡啶结构,该催化剂可利用过氧化氢和烷基过氧化氢实现1,4-醌的顺式双羟基化反应;值得注意的是,研究组首次报道了在顺式双羟基化反应中使用烷基过氧化氢作为末端氧化剂的实例。该体系可将多种1,4-醌高效氧化为顺式-1,2-二醇,产物具有合成价值的产率、优异的化学选择性、区域选择性和对映选择性(高达99% ee),并兼具完全的非对映选择性。
实验与计算化学的机理研究证实,高亲电性的锰(V)-氧-氢氧物种是活性顺式双羟基化中间体,但根据氢过氧化物氧化剂的不同,高价锰-氧中间体的形成机制存在差异。此外,该研究提出了锰(V)-氧-氢氧物种介导的不对称顺式双羟基化两步机理:首先是Re面选择性氧原子进攻位阻较小的C=C键,随后发生羟基回弹,立体化学由初始氧原子进攻步骤决定。该研究首次阐明了通过过氧化氢和烷基过氧化氢氧化生成的高价锰-氧物种介导1,4-醌不对称顺式双羟基化的仿生氧化策略。
附:英文原文
Title: Catalytic Asymmetric cis-Dihydroxylation of Quinones Enabled by a Functional Mimic of Rieske Dioxygenases
Author: Shixin Zheng, Huajie Zhao, Jie Chen, Peng Wu, Binju Wang, Wonwoo Nam, Bin Wang
Issue&Volume: March 18, 2026
Abstract: The development of efficient and practical catalytic asymmetric cis-dihydroxylation reactions using earth-abundant metal catalysts remains a major challenge; the cis-dihydroxylation of 1,4-quinones represents a vital gateway to enantioenriched cis-1,2-diols, which are privileged scaffolds for bioactive natural products and pharmaceuticals. Inspired by the asymmetric cis-dihydroxylation of aromatic and olefinic C═C bonds catalyzed by Rieske dioxygenases, we present a biomimetic strategy for the asymmetric cis-dihydroxylation of 1,4-quinones. Herein, we report an inexpensive and readily accessible nonheme manganese complex, supported by a tetradentate aminopyridine ligand featuring a rigid chiral diamine backbone and a sterically bulky triisopropylsilyl group, for the cis-dihydroxylation of 1,4-quinones by hydrogen peroxide and alkyl hydroperoxides; it is noted that this study reports the first example of using alkyl hydroperoxides as a terminal oxidant in cis-dihydroxylation reactions. Various 1,4-quinones were efficiently oxidized to cis-1,2-diols in synthetically useful yields with excellent chemo-, regio-, and enantioselectivity (up to 99% ee), along with complete diastereoselectivity. Mechanistic evidence from experimental and computational studies supports a highly electrophilic manganese(V)-oxo-hydroxo species as the active cis-dihydroxylating intermediate, although the formation mechanism of the high-valent manganese-oxo intermediate is different depending on the hydroperoxide oxidants. Moreover, a two-step mechanism is proposed for the manganese(V)-oxo-hydroxo-mediated asymmetric cis-dihydroxylation, such as the Re-face selective oxo attack on the less hindered C═C bond, followed by OH-rebound, with stereochemistry dictated by the initial oxo attack. Thus, this study delineates the first biomimetic oxidation strategy enabling asymmetric cis-dihydroxylation of 1,4-quinones mediated by a high-valent manganese-oxo species generated in the reactions of hydrogen peroxide and alkyl hydroperoxides.
DOI: 10.1021/jacs.5c20048
Source: https://pubs.acs.org/doi/abs/10.1021/jacs.5c20048
JACS:《美国化学会志》,创刊于1879年。隶属于美国化学会,最新IF:16.383
官方网址:https://pubs.acs.org/journal/jacsat
投稿链接:https://acsparagonplus.acs.org/psweb/loginForm?code=1000
