耶鲁医学院Nenad Sestan团队宣布他们的最新研究提出了哺乳动物新皮层基因调控网络的适应性进化。该研究于2026年3月18日发表于国际一流学术期刊《自然》杂志上。
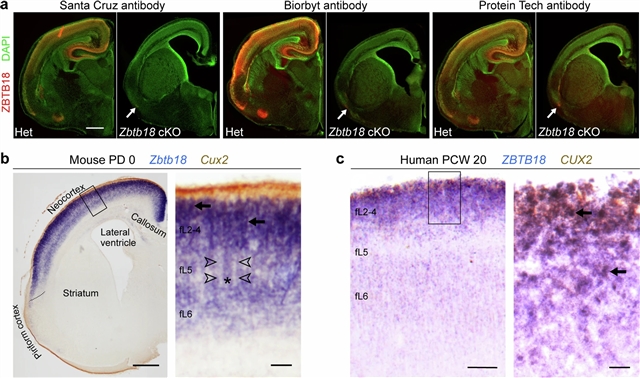
通过比较小鼠ExN亚型的基因表达和推测的顺式调控元件(CREs),并通过跨物种检查,小组确定了哺乳动物特异性的CREs,包括一个由转录因子ZBTB18(也称为RP58、ZFP238或ZNF238)结合的亚群,并与定义脑内和脑外亚型和连性的基因相关,这些基因与智力残疾和自闭症有关。小鼠ExNs中Zbtb18的缺失导致靶基因表达失调,分子多样性降低,皮质-脊髓和胼胝体投射减少,大脑内皮质-皮质关联到前额皮质的投射增加,因此与非哺乳动物的大脑相似。ZBTB18结合基序在胼胝体突起的脑外偏倚推定CREs中高度富集,并在哺乳动物中表现出更高的保护性。这项研究揭示了新皮质外显子网络身份和连接的关键组成部分和哺乳动物特异性进化适应的调控节点。
据介绍,哺乳动物已经进化出一个更复杂的大脑,例如,祖先的兴奋性投射神经元(ExN)的单层背皮层转变为多层大脑新皮层,丰富了各种脑内和脑外ExN亚型,从而建立了专门的投射系统,增强了大脑的连通性和功能。这与现代爬行动物和鸟类的ExNs的单层或伪层柱状组织形成鲜明对比。然而,这些哺乳动物特异性适应的机制仍然是未知的。
附:英文原文
Title: Adaptive evolution of gene regulatory networks in mammalian neocortex
Author: Li, Zhuo, Kaur, Navjot, Santpere, Gabriel, Muchnik, Sydney K., Sindhu, Suvimal Kumar, Qi, Cai, Shibata, Mikihito, Clment, Olivier, Klari, Thomas S., de Martin, Xabier, Luria, Victor, Cho, Hyesun, Li, Mingfeng, Shibata, Akemi, Choi, Sang-Hun, Kim, Hyojin, Tebbenkamp, Andrew T. N., Ma, Shaojie, Han, Wenqi, Kim, Suel-Kee, Pochareddy, Sirisha, Duy, Phan Q., Xing, Xiaojun, Bao, Yunhua, Xu, Xuming, Gladwyn-Ng, Ivan Enghian, Cullen, Hayley Daniella, Paolino, Annalisa, Fenlon, Laura R., Kozulin, Peter, Surez, Rodrigo, Risgaard, Ryan D., Gulden, Forrest O., Karger, Amir, Suzuki, Ikuo K., Hirata, Tatsumi, Gobeske, Kevin T., Richards, Linda J., Sousa, Andr M. M., Heng, Julian I.-T., Sestan, Nenad
Issue&Volume: 2026-03-18
Abstract: Mammals have evolved a more complex brain, exemplified by the transformation of the single-layer dorsal cortex of excitatory projection neurons (ExNs) in ancestors into a multilayered cerebral neocortex1,2,3,4 enriched with diverse intratelencephalic and extratelencephalic ExN subtypes5,6,7, thereby establishing specialized projection systems that enhance brain connectivity and functionality5,6,7,8. This is in contrast to modern reptiles and birds with single-layered or pseudolayered columnar organization of ExNs4,9,10,11,12. However, the mechanisms underlying these mammalian-specific adaptations remain elusive. By comparing the landscape of gene expression and putative cis-regulatory elements (CREs) in mouse ExN subtypes and through cross-species examination, we identified mammalian-specific CREs, including a subset bound by the transcription factor ZBTB18 (also RP58, ZFP238 or ZNF238) and associated with genes defining intratelencephalic and extratelencephalic subtypes and connectivity, which have been implicated in intellectual disability and autism. Deletion of Zbtb18 in mouse ExNs dysregulated target gene expression, reduced molecular diversity, diminished cortico-spinal and callosal projections and increased intrahemispheric cortico-cortical association projections to the prefrontal cortex, thereby resembling non-mammalian brain. ZBTB18 binding motifs are highly enriched in callosally projecting intratelencephalic-biased putative CREs and show higher conservation specifically in mammals. This study uncovers critical components and mammalian-specific evolutionary adaptations within a regulatory node essential for neocortical ExN identity and connectivity.
DOI: 10.1038/s41586-026-10226-y
Source: https://www.nature.com/articles/s41586-026-10226-y
Nature:《自然》,创刊于1869年。隶属于施普林格·自然出版集团,最新IF:69.504
官方网址:http://www.nature.com/
投稿链接:http://www.nature.com/authors/submit_manuscript.html
