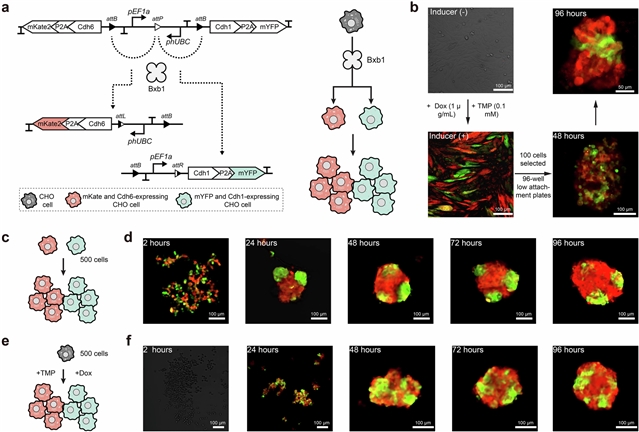
细胞比例控制的合成电路,这一成果由中国科学院深圳先进技术研究院钟超小组经过不懈努力而取得。该项研究成果发表在2026年3月18日出版的《自然》上。
在这里,该研究团队介绍了一套重组酶介导的遗传装置,旨在准确控制群体比例,使不同的功能分布在多种细胞类型。该研究团队系统地评估了影响重组效率的关键参数,并开发了数据驱动模型来可靠地预测二元分化结果。使用这些器件,研究团队构建了并联和串联电路拓扑来实现热定义的多步细胞命运分支程序。分支装置促进了从单一创始酵母菌株的精密发酵财团的自主分化,优化了诸如色素沉着和纤维素降解等应用的细胞类型比例。在哺乳动物细胞中也获得了类似的效果。研究组还通过细胞粘附分子协调自组织,设计了具有遗传编码形态的多细胞聚集体。他们的工作提供了基于重组酶的细胞命运分支机制的全面表征,并介绍了构建合成联合体和多细胞组装的方法。
研究人员表示,基因工程的最新进展为人工分化原核和真核细胞群提供了多种工具。然而,如何精确控制由单一奠基者产生的群体中多种细胞类型的比例仍然是一个重大挑战。
附:英文原文
Title: Synthetic circuits for cell ratio control
Author: An, Bolin, Tang, Tzu-Chieh, Zhang, Qian, Wang, Teng, Wang, Yanyi, Gan, Kesheng, Liu, Kun, Zhang, Daniel L., Liu, Yuzhu, Pan, Yu Kui, Yu, Min, Shaw, William M., Liang, Qianyi, Wang, Yaomin, Vaiana, Christopher A., Lou, Chunbo, Voigt, Christopher A., Lu, Timothy K., Church, George M., Zhong, Chao
Issue&Volume: 2026-03-18
Abstract: Recent advances in genetic engineering have provided diverse tools for artificially diversifying both prokaryotic and eukaryotic cell populations1,2,3,4,5,6. However, achieving precise control over the ratios of multiple cell types within a population derived from a single founder remains a major challenge. Here we introduce a suite of recombinase-mediated genetic devices designed to accurately control population ratios, enabling the distribution of distinct functionalities across multiple cell types. We systematically evaluated key parameters that influence recombination efficiency and developed data-driven models to reliably predict binary differentiation outcomes. Using these devices, we constructed parallel and series circuit topologies to implement user-defined, multistep cell-fate branching programs. The branching devices facilitated the autonomous differentiation of precision fermentation consortia from a single founder yeast strain, optimizing cell-type ratios for applications such as pigmentation and cellulose degradation. Similar effects were obtained with mammalian cells. We also engineered multicellular aggregates with genetically encoded morphologies by coordinating self-organization through cell adhesion molecules. Our work provides a comprehensive characterization of recombinase-based cell-fate branching mechanisms and introduces an approach for constructing synthetic consortia and multicellular assemblies.
DOI: 10.1038/s41586-026-10259-3
Source: https://www.nature.com/articles/s41586-026-10259-3
Nature:《自然》,创刊于1869年。隶属于施普林格·自然出版集团,最新IF:69.504
官方网址:http://www.nature.com/
投稿链接:http://www.nature.com/authors/submit_manuscript.html
