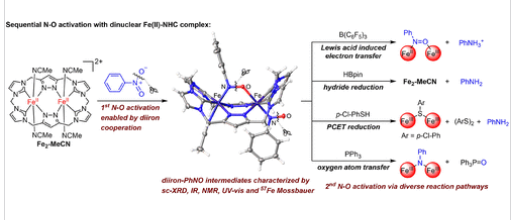
近日,清华大学章名田团队解读了硝基芳烃的逐步N-O活化:铁硝基芳烃中间体的电子结构和反应性。这一研究成果于2026年3月17日发表在《美国化学会志》上。
硝基化合物(包括NOx-和含硝基有机底物)还原转化过程中的N–O键活化,因其在生物系统和合成化学领域的重要意义而备受关注。硝基的还原转化涉及连续的N–O键断裂过程,其中亚硝基芳烃(ArNO)作为关键中间体却常难以捕获。尽管铁是参与这类过程的生物及合成相关金属,但其作用机制,特别是多核协同效应的理解仍不完善。
研究组报道了双核铁N-杂环卡宾(NHC)配合物Fe2-MeCN能够实现硝基苯的连续N–O键活化,进而分离并详细表征了两个铁-亚硝基芳烃中间体Fe2-PhNO和Fe2-(PhNO)2。通过光谱学、晶体学研究结合DFT计算,鉴定出极化N–O基团的强电荷转移相互作用。
这种电子结构支撑了多样化的N–O活化路径,包括路易斯酸诱导电子转移、氢化物还原、质子耦合电子转移(PCET)及氧原子转移。化学计量反应性研究表明,配位ArNO的亲核氧可被亲电试剂有效攻击,而与亲核试剂的反应则需要更剧烈的条件。最后,Fe2-MeCN可作为将硝基芳烃还原为芳胺的高效催化剂,并促进前所未有的铁介导杂环化反应,凸显了双金属协同作用在此多功能反应性中的关键作用。
附:英文原文
Title: Deciphering the Stepwise N–O Activation of Nitroarenes: Electronic Structure and Reactivity of Iron-Nitrosoarene Intermediates
Author: Xinyu Xu, Kai Hua, Fei Xie, Ming-Tian Zhang
Issue&Volume: March 17, 2026
Abstract: N–O bond activation in reductive transformation of both NOx– and organic substrates with nitro functional groups has garnered significant interest because of its significance in both biological systems and synthetic chemistry. The reductive transformation of nitro groups involves sequential N–O bond cleavages, with nitrosoarenes (ArNO) as key yet often elusive intermediates. While iron is a biologically and synthetically relevant metal for such processes, the mechanistic understanding, particularly the role of multinuclear cooperation, remains underdeveloped. Herein, we report that a dinuclear iron N-heterocyclic carbene (NHC) complex, Fe2-MeCN, enables the sequential N–O bond activation of nitrobenzene, leading to the isolation and detailed characterization of two iron–nitrosoarene intermediates, Fe2-PhNO and Fe2-(PhNO)2. Spectroscopic and crystallographic studies, complemented by DFT calculations, identified strong charge-transfer interactions that polarize the N–O moiety. This electronic structure underpins a diverse suite of N–O activation pathways, including Lewis acid-induced electron transfer, hydride reduction, proton-coupled electron transfer (PCET), and oxygen atom transfer. The stoichiometric reactivity reveals that the nucleophilic oxygen of the coordinated ArNO can be efficiently attacked by electrophiles, while reactions with nucleophiles require more forcing conditions. Finally, Fe2-MeCN functions as a competent catalyst for the reduction of nitroarenes to anilines and facilitates unprecedented iron-mediated heterocyclizations, underscoring the critical role of bimetallic cooperativity in this multifunctional reactivity.
DOI: 10.1021/jacs.5c19714
Source: https://pubs.acs.org/doi/abs/10.1021/jacs.5c19714
JACS:《美国化学会志》,创刊于1879年。隶属于美国化学会,最新IF:16.383
官方网址:https://pubs.acs.org/journal/jacsat
投稿链接:https://acsparagonplus.acs.org/psweb/loginForm?code=1000
