普林斯顿大学Martin C. Jonikas小组近日取得一项新成果。经过不懈努力,他们揭示了激酶KEY1通过破坏相分离相互作用在整个细胞周期中调控蛋白核凝聚体的大小。2026年3月17日出版的《自然—细胞生物学》杂志发表了这项成果。
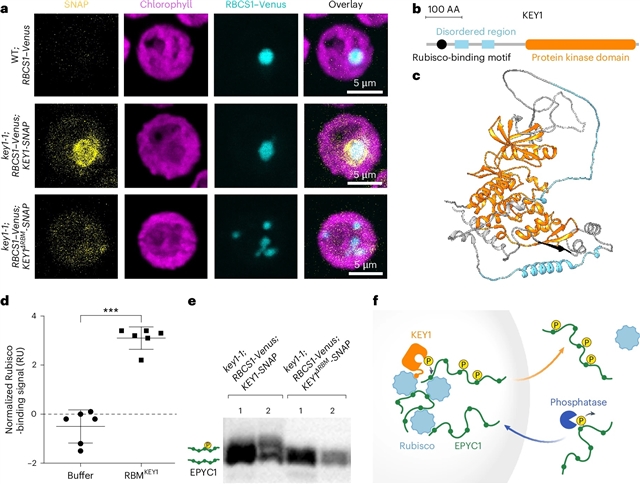
该课题组人员在模型藻莱茵衣藻(Chlamydomonas reinhardtii)中发现了一种激酶KEY1,它在整个细胞周期中调节类pyrenoid凝聚物的大小和数量动态,是正常的类pyrenoid功能和生长所必需的。与野生型不同,KEY1突变细胞在整个细胞周期中有多个较小的凝聚体,在细胞分裂过程中不能溶解。研究团队发现KEY1定位于凝聚物,并通过破坏凝聚物核心成分(二氧化碳固定酶Rubisco及其连接蛋白EPYC1)之间的相互作用,通过EPYC1磷酸化促进凝聚物的溶解。该研究组建立了一个生物物理模型,概括了KEY1介导的凝析物大小和数量调节,并提出了控制凝析物位置的机制。这些数据为了解类芘和其他生物分子凝聚物的大小、数目、位置和溶解调节的机理奠定了基础。
据介绍,生物分子凝聚体在空间上组织细胞功能,但其大小、数量、溶解和再凝聚的调控机制尚不清楚。pyrenoid是一种藻类生物分子凝结物,介导了全球三分之一的二氧化碳固定,通常每个叶绿体存在一个大的凝结物,但在细胞分裂过程中,它会短暂溶解并重新配置成多个较小的凝结物。
附:英文原文
Title: Kinase KEY1 controls pyrenoid condensate size throughout the cell cycle by disrupting phase separation interactions
Author: He, Shan, Lemma, Linnea M., Martinez-Calvo, Alejandro, He, Guanhua, Hennacy, Jessica H., Wang, Lianyong, Ergun, Sabrina L., Rai, Ashwani K., Wang, Colton, Bunday, Luke, Kayser-Browne, Angelo, Wang, Quan, Brangwynne, Clifford P., Wingreen, Ned S., Jonikas, Martin C.
Issue&Volume: 2026-03-17
Abstract: Biomolecular condensates spatially organize cellular functions, but the regulation of their size, number, dissolution and re-condensation is poorly understood. The pyrenoid, an algal biomolecular condensate that mediates one-third of global CO2 fixation, typically exists as one large condensate per chloroplast, but during cell division it transiently dissolves and reconfigures into multiple smaller condensates. Here, we identify a kinase, KEY1, in the model alga Chlamydomonas reinhardtii that regulates pyrenoid condensate size and number dynamics throughout the cell cycle and is necessary for normal pyrenoid function and growth. Unlike the wild type, key1 mutant cells have multiple smaller condensates throughout the cell cycle that fail to dissolve during cell division. We show that KEY1 localizes to the condensates and promotes their dissolution by disrupting interactions between their core constituents, the CO2-fixing enzyme Rubisco and its linker protein EPYC1, through EPYC1 phosphorylation. We develop a biophysical model that recapitulates KEY1-mediated condensate size and number regulation and suggests a mechanism for controlling condensate position. These data provide a foundation for the mechanistic understanding of the regulation of size, number, position and dissolution in pyrenoids and other biomolecular condensates.
DOI: 10.1038/s41556-026-01908-w
Source: https://www.nature.com/articles/s41556-026-01908-w
Nature Cell Biology:《自然—细胞生物学》,创刊于1999年。隶属于施普林格·自然出版集团,最新IF:28.213
官方网址:https://www.nature.com/ncb/
投稿链接:https://mts-ncb.nature.com/cgi-bin/main.plex
