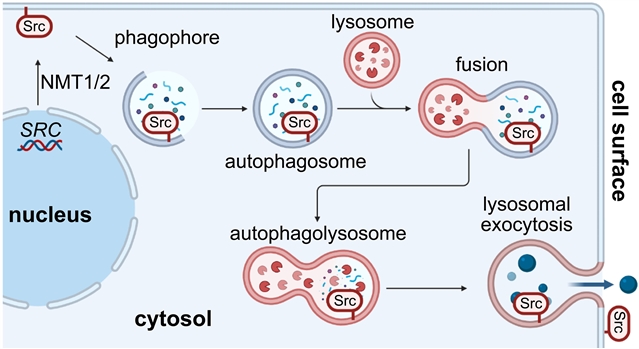
在这项工作中,该研究组发现Src在体外和体内都是非典型易位和倒置到癌症细胞表面上。该研究组发现自噬溶酶体胞吐(ALE)是一种在癌细胞系中突出的分泌机制。Src代表了通过这一过程运输的膜锚定蛋白家族的典型例子。
此外,在原发肿瘤中发现了这种细胞外膜相关Src (eSrc),在细胞培养系统和同种异种移植模型中,基于抗Src抗体的疗法介导了肿瘤细胞的杀伤。它们是细胞内N-肉豆油酰基化蛋白,典型的是Src,可以在癌症中拓扑倒置到细胞表面,并以抗体治疗为目标。
据了解,原癌基因Src的过表达在多种癌症中都很常见。
附:英文原文
Title: Autophagolysosomal exocytosis inverts Src kinase onto the cell surface in cancer
Author: Corleone S. Delaveris, Rita P. Loudermilk, Apurva Pandey, Soumya G. Remesh, Trenton M. Peters-Clarke, Snehal D. Ganjave, William J. N. Dougherty, Henry M. Delavan, Chunyue Wang, Jesse Ling, Juan Antonio Camara Serrano, Fernando Salangsang, Chien-Kuang Cornelia Ding, Nancy Greenland, Carissa E. Chu, Sima Porten, Veronica Steri, Jonathan Chou, Michael J. Evans, Kevin K. Leung, James A. Wells
Issue&Volume: 2026-03-12
Abstract: Overexpression of the proto-oncogene Src is common to a wide variety of cancers. In this work, we found that Src is noncanonically translocated and inverted onto the cell surface in cancer, both in vitro and in vivo. We identified autophagolysosomal exocytosis (ALE) as a secretory mechanism prominent in cancer cell lines. Src represents the prototypical example of a family of membrane-anchored proteins that are transported by this process. Furthermore, this extracellular membrane–associated Src (eSrc) was found in primary tumors, and anti-Src antibody-based therapies mediated tumor cell killing in cell culture systems and in mouse xenograft models. Thus, intracellular N-myristoylated proteins, prototypically Src, can be topologically inverted onto the cell surface in cancer and targeted with antibody therapeutics.
DOI: aec1778
Source: https://www.science.org/doi/10.1126/science.aec1778
