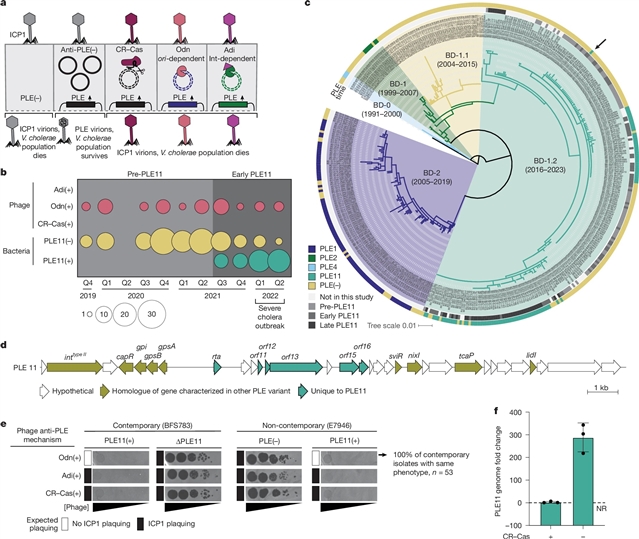
加州大学Kimberley D. Seed团队取得一项新突破。他们的论文发现了通过临床监测捕捉噬菌体-病原体共同进化的动态。相关论文于2026年3月11日发表在《自然》杂志上。
通过在霍乱流行的孟加拉国进行临床监测,该课题组人员获得了一种寄生抗噬菌体移动基因元件PLE11,该基因元件在最近有记录以来最大的霍乱暴发期间启动了选择性清除。PLE11对共循环ICP1表现出强大的抗噬菌体活性,这解释了其快速和主导的出现。该课题组人员确定了PLE11编码的Rta作为防御负责,并提供了Rta限制噬菌体尾部组装的证据。
通过实验进化,研究小组预测了噬菌体对PLE11的反适应性,并记录了临床ICP1的最终出现和选择,从而实现了趋同的进化结果。最后,研究团队发现了在Rta限制噬菌体尾部组装的情况下,PLE如何平衡其对ICP1尾部蛋白的水平传播依赖:PLE构建由移动遗传元件编码蛋白和噬菌体编码蛋白组成的嵌合尾部,以确保其传播。总的来说,他们的发现揭示了在临床相关背景下全球重要病原体及其病毒自然选择的分子基础。
据了解,细菌利用多种防御系统来抵御噬菌体的捕食,其中许多防御系统都编码在水平传播的可移动遗传元件上。反过来,噬菌体进化出对抗防御,推动了一场动态的军备竞赛,但在人类疾病背景下,这一竞赛仍未得到充分探索。对于腹泻病原体霍乱弧菌,患者粪便中ICP1溶解噬菌体负担较高与疾病严重程度降低相关。然而,裂解噬菌体驱动霍乱弧菌选择的直接分子证据尚未得到证实。
附:英文原文
Title: Capturing dynamic phage–pathogen coevolution by clinical surveillance
Author: Mathur, Yamini, Boyd, Caroline M., Farnham, Jeannette E., Monir, Md Mamun, Islam, Mohammad Tarequl, Sultana, Marzia, Ahmed, Tahmeed, Alam, Munirul, Seed, Kimberley D.
Issue&Volume: 2026-03-11
Abstract: Bacteria harness diverse defence systems that protect against phage predation1, many of which are encoded on horizontally transmitted mobile genetic elements2. In turn, phages evolve counter-defences3, driving a dynamic arms race that remains underexplored in human disease contexts. For the diarrhoeal pathogen Vibrio cholerae, a higher burden of its lytic phage ICP1 in patient stool correlates with reduced disease severity4. However, direct molecular evidence of lytic phages driving selection of epidemic V. cholerae has not been demonstrated. Here, through clinical surveillance in cholera-endemic Bangladesh, we capture the acquisition of a parasitic anti-phage mobile genetic element, PLE11, that initiated a selective sweep coinciding with the largest cholera outbreak in recent records. PLE11 showed potent anti-phage activity against cocirculating ICP1, explaining its rapid and dominating emergence. We identify PLE11-encoded Rta as the defence responsible and provide evidence that Rta restricts phage tail assembly. Using experimental evolution, we predict phage counteradaptations against PLE11 and document the eventual emergence and selection of clinical ICP1 that achieve a convergent evolutionary outcome. Finally, we discover how PLEs balance their dependence on ICP1 tail proteins for horizontal transmission with the restriction of phage tail assembly by Rta: PLEs construct chimeric tails composed of both mobile genetic element-encoded and phage-encoded proteins to ensure their transmission. Collectively, our findings reveal the molecular basis of the natural selection of a globally important pathogen and its virus in a clinically relevant context.
DOI: 10.1038/s41586-026-10136-z
Source: https://www.nature.com/articles/s41586-026-10136-z
Nature:《自然》,创刊于1869年。隶属于施普林格·自然出版集团,最新IF:69.504
官方网址:http://www.nature.com/
投稿链接:http://www.nature.com/authors/submit_manuscript.html
