东京大学Hideaki E. Kato近日取得一项新成果。经过不懈努力,他们研究出GPCR识别和激活G蛋白的动态基础。2026年3月11日出版的《自然》杂志发表了这项成果。
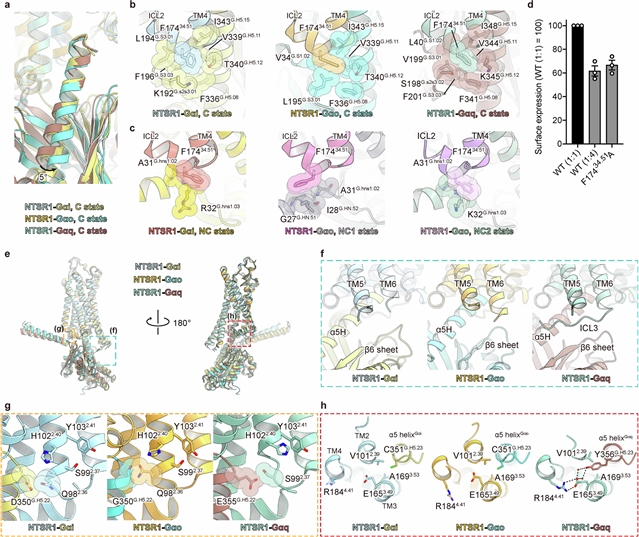
在这里,该课题组展示了具有最小修饰Go和Gq的人类神经紧张素受体1型(NTSR1)的低温电子显微镜结构,显示了受体的细胞内表面如何动态重排以适应每种G蛋白亚型。
此外,NTSR1-Gi的时间分辨冷冻电镜分析显示了G蛋白在GDP/GTP结合上的解离过程。对20多个中间体进行了表征,并辅以突变和计算分析,确定了它们的关键机制特征。首先,GDP/GTP诱导Gi从典型和非典型活性构象释放,具有不同的动力学。其次,NTSR1介导常见的细胞内重排,以识别不同的G蛋白亚型,并促进单一亚型的激活。第三,与Gβγ的分离涉及到Gα开关I-III的逐步重塑。最后,Gi通过与Gs不同的途径与受体解离,规范和非规范的NTSR1-Gi复合物在解离轨迹上进一步分化。这些发现为理解GPCR信号动力学和指导信号靶向治疗发展提供了一个全面的框架。
研究人员表示,G蛋白偶联受体(GPCR)信号通过异三聚体G蛋白发生,其选择性激活导致不同的细胞结果。虽然已经确定了200多个GPCR-G蛋白复合物结构,但这些静态快照对G蛋白结合和解离的动力学提供了有限的见解。
附:英文原文
Title: The dynamic basis of G-protein recognition and activation by a GPCR
Author: Kobayashi, Kazuhiro, Kawakami, Kouki, Matsui, Toshiki E., Yokoi, Shun, Fukuda, Masahiro, Narita, Tomohiro J., Arai, Hiroki, Tambo, Mai, Sumikama, Takashi, Tatsumi, Manae, Yamashita, Keitaro, Koyanagi, Junki, Kugawa, Mai, Ikeda, Hisako, Sumino, Ayumi, Mitsutake, Ayori, Kobilka, Brian K., Inoue, Asuka, Kato, Hideaki E.
Issue&Volume: 2026-03-11
Abstract: G-protein-coupled receptor (GPCR) signalling occurs through heterotrimeric G proteins, whose selective activation leads to distinct cellular outcomes1. Although more than 200 GPCR–G protein complex structures have been determined2, these static snapshots provide limited insight into the dynamics of G-protein association and dissociation. Here we present cryo-electron microscopy structures of human neurotensin receptor type 1 (NTSR1) with minimally modified Go and Gq, showing how the receptor’s intracellular surface dynamically rearranges to accommodate each G-protein subtype. Furthermore, time-resolved cryo-electron microscopy analyses of NTSR1–Gi visualized G-protein dissociation processes on GDP/GTP binding. Characterization of more than 20 intermediates, complemented by mutational and computational analyses, identifies four key mechanistic features. First, GDP/GTP induces Gi release from both canonical and non-canonical active conformations with distinct kinetics. Second, NTSR1 uses common intracellular rearrangements to recognize different G-protein subtypes and to promote activation of a single subtype. Third, separation from Gβγ involves stepwise remodelling of the Gα switches I–III. Finally, Gi dissociates from the receptor through a pathway that is distinct from that of Gs, and the canonical and non-canonical NTSR1–Gi complexes further diverge in their dissociation trajectories. These findings provide a comprehensive framework for understanding GPCR signalling dynamics and guiding signal-targeted therapeutic development.
DOI: 10.1038/s41586-026-10228-w
Source: https://www.nature.com/articles/s41586-026-10228-w
Nature:《自然》,创刊于1869年。隶属于施普林格·自然出版集团,最新IF:69.504
官方网址:http://www.nature.com/
投稿链接:http://www.nature.com/authors/submit_manuscript.html
