加州大学James A. Olzmann团队的研究显示,CLCC1促进肝中性脂质通量和核孔复合物组装。相关论文于2026年2月25日发表在《自然》杂志上。
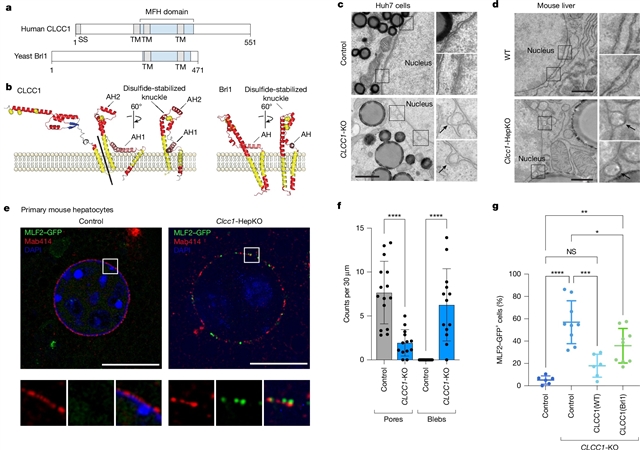
在这里,课题组在不同的代谢状态下进行了一系列CRISPR-Cas9筛选,最终鉴定出CLCC1是肝细胞中性脂质储存和分泌的关键调节因子。CLCC1缺失导致肝癌细胞形成大脂滴,CLCC1敲除导致小鼠肝脂肪变性。脂滴存在于Clcc1敲除肝细胞内质网的管腔中,并表现出脂蛋白的特性,表明中性脂通量发生了深刻的变化。CLCC1的缺失也导致核膜疝的积累,并伴有核孔的减少。远程同源性搜索发现CLCC1中的一个结构域与酵母Brl1和Brr6同源,这两个因子在核孔复合物组装过程中促进核膜的融合。分子动力学模拟和诱变研究支持CLCC1介导膜弯曲和膜离子的模型。研究人员提出CLCC1介导膜离子促进肝中性脂质通量和核孔复合物组装。
据介绍,脂质储存和分泌失衡导致肝脂肪变性,肝细胞中脂滴积聚。他们对控制肝细胞中性脂向胞质脂滴或分泌脂蛋白通道的机制的理解仍然不完整。
附:英文原文
Title: CLCC1 promotes hepatic neutral lipid flux and nuclear pore complex assembly
Author: Mathiowetz, Alyssa J., Meymand, Emily S., Parlakgl, Gne, van Hilten, Niek, Torres, Emily F., Artico, Leonardo L., Deol, Kirandeep K., Lange, Mike, Pang, Stephany P., Doubravsky, Cody E., Roberts, Melissa A., Jorgens, Danielle M., Zalpuri, Reena, Kang, Misun, Boone, Casadora, Parks, Brian W., Zhang, Yaohuan, Morgens, David W., Newman, Emily Tso, Zhou, Yingjiang, Talukdar, Saswata, Grabe, Michael, Ku, Gregory, Levine, Tim P., Arruda, Ana Paula, Olzmann, James A.
Issue&Volume: 2026-02-25
Abstract: Imbalances in lipid storage and secretion lead to hepatic steatosis, the accumulation of lipid droplets in hepatocytes1,2. Our understanding of the mechanisms that govern the channelling of neutral lipids in hepatocytes towards cytosolic lipid droplets or secreted lipoproteins remains incomplete3,4. Here we performed a series of CRISPR–Cas9 screens under different metabolic states that led to the identification of CLCC1 as a critical regulator of neutral lipid storage and secretion in hepatocytes. Loss of CLCC1 resulted in the buildup of large lipid droplets in hepatoma cells and Clcc1 knockout in mice caused liver steatosis. Lipid droplets were present in the lumen of the endoplasmic reticulum of the Clcc1-knockout hepatocytes and exhibited properties of lipoproteins, indicating a profound shift in neutral lipid flux. The loss of CLCC1 also led to the accumulation of nuclear membrane herniations accompanied by a reduction in nuclear pores. Remote homology searches identified a domain in CLCC1 that is homologous to yeast Brl1 and Brr6, factors that promote nuclear envelope fusion during nuclear pore complex assembly. Molecular dynamics simulations and mutagenesis studies support a model in which CLCC1 mediates membrane bending and fusion. We propose that CLCC1 mediates membrane fusion to promote hepatic neutral lipid flux and nuclear pore complex assembly.
DOI: 10.1038/s41586-025-10064-4
Source: https://www.nature.com/articles/s41586-025-10064-4
Nature:《自然》,创刊于1869年。隶属于施普林格·自然出版集团,最新IF:69.504
官方网址:http://www.nature.com/
投稿链接:http://www.nature.com/authors/submit_manuscript.html
