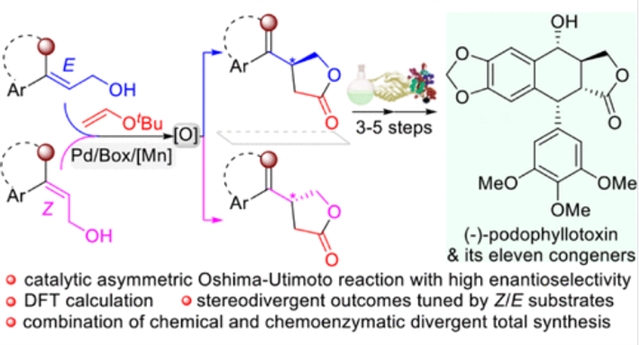
近日,兰州大学张辅民团队研究了通过不对称Oshima-Utimoto反应催化β-取代γ-内酯的对映选择性构建(-)-鬼臼毒素及其11个同系物的分散全合成。该研究于2025年8月19日发表在《美国化学会杂志》上。
研究组报道了第一个高度对映选择性的Oshima-Utimoto反应,该反应与随后的一锅氧化相结合,从两种化学原料(烯丙醇和乙烯基醚)中合成了具有高对映选择性(高达97%ee)的重要对映富集β-取代γ-内酯。值得注意的是,通过策略性地选择Z-或E-烯丙醇底物,可以精确控制手性β-取代的γ-内酯的绝对构型,从而提供了一种获取手性γ-内酯对映异构体的替代方法。
利用这种催化不对称转化作为关键步骤,研究组通过化学和化学酶策略的结合,从商业材料中仅用6-8步就实现了天然芳基四氢萘木脂素药物(-)-鬼臼毒素及其11种结构同系物的简洁立体发散全合成。该方案以开发催化不对称Oshima-Utimoto反应为特征,不仅为合成有价值的β-取代的γ-内酯提供了一条简洁的路线,而且通过对合成前体进行轻微修饰,为制备结构多样的天然药物(-)-鬼臼毒素衍生物建立了一个潜在的平台。
附:英文原文
Title: Enantioselective Construction of β-Substituted γ-Lactone through Catalytic Asymmetric Oshima–Utimoto Reaction: Divergent Total Syntheses of ()-Podophyllotoxin and Its Eleven Congeners
Author: Zhi-Juan He, Ma-Haoran Hong, Ling-Hui Zhang, Wen-Hui Xiong, Fu-Min Zhang
Issue&Volume: August 19, 2025
Abstract: We report the first highly enantioselective Oshima–Utimoto reaction, which is combined with subsequent one-pot oxidation to construct synthetically important enantioenriched β-substituted γ-lactones with high enantioselectivity (up to 97% ee) from two chemical feedstocks (allylic alcohols and vinyl ethers). Significantly, the absolute configuration of chiral β-substituted γ-lactones can be precisely controlled by strategically selecting Z- or E-allylic alcohol substrates, thereby providing an alternative access to both enantiomers of chiral γ-lactones. Capitalizing on this catalytic asymmetric transformation as a key step, we have achieved a concise and stereodivergent total synthesis of the natural aryltetralin lignan drug ()-podophyllotoxin and its 11 structural congeners in only 6–8 steps from commercial materials through the combination of chemical and chemoenzymatic strategies. The present protocol, featuring the development of a catalytic asymmetric Oshima–Utimoto reaction, not only offers a concise route to the synthetically valuable β-substituted γ-lactones, but also establishes a potential platform for preparing structurally diverse natural drug ()-podophyllotoxin derivatives through minor modifications of synthetic precursors.
DOI: 10.1021/jacs.5c07358
Source: https://pubs.acs.org/doi/abs/10.1021/jacs.5c07358
JACS:《美国化学会志》,创刊于1879年。隶属于美国化学会,最新IF:16.383
官方网址:https://pubs.acs.org/journal/jacsat
投稿链接:https://acsparagonplus.acs.org/psweb/loginForm?code=1000
