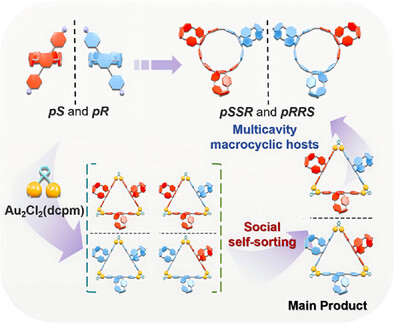
近日,浙江大学华彬团队研究了具有圆偏振发光的[9]环对苯乙烯柱[5]芳烃杂手性多腔大环三聚体的手性自分选合成。该项研究成果发表在2025年8月11日出版的《德国应用化学》杂志上。
基于柱[n]芳烃的立体化学控制多腔大环主体的精确合成是超分子化学领域的一个重大挑战。这一困难主要源于在合成过程中自发产生的平面手性诱导的立体异构体混合物,这使分离过程相当复杂。
研究组引入了一种利用三角形金配合物的手性自分选策略,使[9]环对苯乙烯-柱[5]芳烃三聚体([9]CPP-3P[5] a)的立体化学控制合成成为可能。传统的合成方法通常产生柱芳烃三聚体的立体异构体(pSSS、pRRR、pSSR和pRRS),与之不同的是,该方法具有选择性,主要产生[9]CPP-3P[5]a的pSSR和pRRS异构体,只有微量的pSSS和pRRR异构体。
晶体学分析表明,这种立体选择性是由于在六方配合物组装过程中Au─C σ-键的动态重组,与特定的磷化氢配体构象和柱间芳烃相互作用协同作用所致。所得到的手性三聚体具有圆极化发光(CPL)特性,利用柱状[5]芳烃单元优越的主客体络合能力,有利于制造具有CPL活性的超分子聚合物,为手性功能材料的开发开辟了新的途径。
附:英文原文
Title: Social Chiral Self-Sorting Synthesis of [9]Cycloparaphenylene-Pillar[5]arene Heterochiral Multicavity Macrocyclic Trimers with Circularly Polarized Luminescence
Author: Shiyu Tan, Ye-Qiang Han, Guangguo Wang, Liya Chen, Li Shao, Mingrui Xiao, Bohan Zhao, Feihe Huang, Bin Hua
Issue&Volume: 2025-08-11
Abstract: The precise synthesis of stereochemically controlled multicavity macrocyclic hosts based on pillar[n]arenes presents a significant challenge in supramolecular chemistry. This difficulty primarily stems from the spontaneous generation of planar chirality-induced stereoisomer mixtures during synthetic procedures, which considerably complicates the isolation process. In this study, we introduce a social chiral self-sorting strategy utilizing a triangular gold complex, enabling the stereochemically controlled synthesis of [9]cycloparaphenylene-pillar[5]arene trimers ([9]CPP-3P[5]A). Diverging from conventional synthetic approaches that typically yield four stereoisomers of pillararene trimers (pSSS, pRRR, pSSR, and pRRS), our methodology demonstrates selectivity, predominantly generating pSSR and pRRS isomers of [9]CPP-3P[5]A, with only trace amounts of pSSS and pRRR isomers. This stereoselectivity, as elucidated by crystallographic analysis, arises from the dynamic reorganization of Au─C σ-bonds during the assembly of the hexagold(I) complex, synergistically combined with specific phosphine ligand conformations and inter-pillararene interactions. The resulting chiral trimers exhibit circularly polarized luminescence (CPL) properties, which facilitates the fabrication of CPL-active supramolecular polymers by leveraging the superior host–guest complexation capability of the pillar[5]arene units, opening new avenues for the development of chiral functional materials.
DOI: 10.1002/anie.202508361
Source: https://onlinelibrary.wiley.com/doi/10.1002/anie.202508361
Angewandte Chemie:《德国应用化学》,创刊于1887年。隶属于德国化学会,最新IF:16.823
官方网址:https://onlinelibrary.wiley.com/journal/15213773
投稿链接:https://www.editorialmanager.com/anie/default.aspx
