中山大学刘建波课题组在研究中取得进展。他们提出了N-单氟甲基酰胺和氨基甲酸酯的制备、分离和储存。相关论文于2025年3月4日发表在《自然—化学》杂志上。
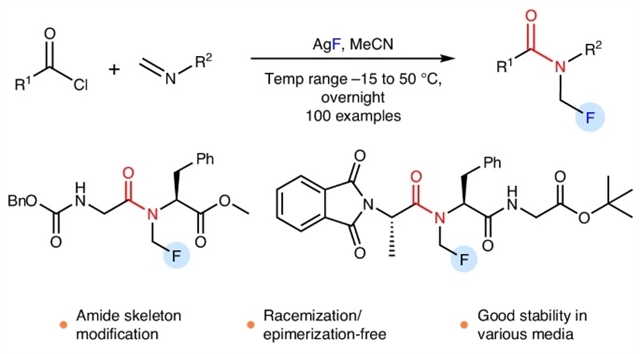
本文报道了用简单、机械的化学方法制备N-CH2F酰胺类和氨基甲酸酯类化合物。N-CH2F酰胺的合成是通过亚胺的连续酰化和氟化实现的,并直接用于药物、肽和杂芳酰胺的修饰,而不需要外消旋化或外映异构化。三乙胺的主题是分离N-CH2F酰胺的关键。9种结构各异的N-CH2F酰胺在8种不同介质中的稳定性测试表明,大多数化合物在24 h内保持60-100%的完整性。
据了解,N-单氟甲基(N-CH2F)酰胺结合了酰胺和单氟甲基基序,代表了酰胺键的实际修饰,可以模拟N-CH3酰胺。尽管N-CH2F在转化多肽和拟肽物方面具有潜在价值,但这种结构的存在一直存在争议。
附:英文原文
Title: Preparation, separation and storage of N-monofluoromethyl amides and carbamates
Author: Tao, Min, Qian, Jiasheng, Deng, Linbei, Wilson, David M., Zhang, Xiangsong, Liu, Jianbo
Issue&Volume: 2025-03-04
Abstract: N-monofluoromethyl (N-CH2F) amides, combining amide and monofluoromethyl motifs, represent a practical modification of the amide bond that can mimic N-CH3 amides. Despite the potential value in transforming peptides and peptidomimetics with N-CH2F, the very existence of this structure has been controversial. Here we report the preparation of N-CH2F amides and carbamates via simple and robust chemical methods. The syntheses of N-CH2F amides were achieved via successive acylation and fluorination of imines and directly used in the modification of drugs, peptides and heteroaryl amides without racemization or epimerization. The use of triethylamine is the key to the separation of N-CH2F amides. The stability of nine structurally diverse N-CH2F amides was tested in eight different media, showing that most compounds remained 60–100% intact for 24h.
DOI: 10.1038/s41557-025-01767-2
Source: https://www.nature.com/articles/s41557-025-01767-2
Nature Chemistry:《自然—化学》,创刊于2009年。隶属于施普林格·自然出版集团,最新IF:24.274
官方网址:https://www.nature.com/nchem/
投稿链接:https://mts-nchem.nature.com/cgi-bin/main.plex
