马德里自治大学Mariola Tortosa小组揭示了对映选择性光催化合成邻二取代苯生物同位酯[2.1.1]。相关论文于2025年2月25日发表在《自然-化学》杂志上。
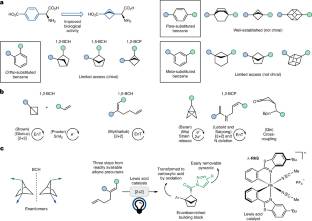
研究团队研制了一种lewis酸催化的[2+ 2]通过光环加成获得这些基序作为对映体富集的支架,为它们结合到各种药物类似物中提供了有效的途径。原药物靶向的特定蛋白质中保留了含双环[2.1.1]己烷的类似物的生物活性,证实了该片段作为邻取代苯环的生物同位体的适用性。
此外,该课题组研究了药物类似物的不同对映体在一组肿瘤细胞系中选择性诱导细胞毒性的潜力,观察到两种对映体的作用明显不同,并且比相应的基于sp2的药物有实质性的改善。这表明药物类似物的绝对构型和三维结构的控制对其生物学特性有很大的影响。
研究人员表示,1,5-二取代双环[2.1.1]己烷是具有明确出口载体的桥接支架,由于它们是邻取代苯环的饱和生物同位异构体,因此在药物化学中越来越受欢迎。
附:英文原文
Title: Enantioselective photocatalytic synthesis of bicyclo[2.1.1]hexanes as ortho-disubstituted benzene bioisosteres with improved biological activity
Author: Garrido-Garca, Pablo, Quirs, Irene, Miln-Rois, Paula, Ortega-Gutirrez, Silvia, Martn-Fontecha, Mar, Campos, Luis A., Somoza, lvaro, Fernndez, Israel, Rigotti, Thomas, Tortosa, Mariola
Issue&Volume: 2025-02-25
Abstract: 1,5-Disubstituted bicyclo[2.1.1]hexanes are bridged scaffolds with well-defined exit vectors that are becoming increasingly popular building blocks in medicinal chemistry because they are saturated bioisosteres of ortho-substituted phenyl rings. Here we have developed a Lewis-acid-catalysed [2+2] photocycloaddition to obtain these motifs as enantioenriched scaffolds, providing an efficient approach for their incorporation in a variety of drug analogues. Retention of the biological activity of the bicyclo[2.1.1]hexane-containing analogues in the specific proteins targeted by the original drugs has confirmed the suitability of this moiety to serve as a bioisostere of ortho-substituted phenyl rings. Moreover, we have studied the potential of the different enantiomers of the drug analogues to selectively induce cytotoxicity in a panel of tumour cell lines, observing markedly differential effects for the two enantiomers and a substantial improvement over the corresponding sp2-based drugs. This showcases that the control of the absolute configuration and tridimensionality of the drug analogue has a large impact on its biological properties.
DOI: 10.1038/s41557-025-01746-7
Source: https://www.nature.com/articles/s41557-025-01746-7
Nature Chemistry:《自然—化学》,创刊于2009年。隶属于施普林格·自然出版集团,最新IF:24.274
官方网址:https://www.nature.com/nchem/
投稿链接:https://mts-nchem.nature.com/cgi-bin/main.plex
