新加坡国立大学Ming Joo Koh团队的一项最新研究报道了催化插入二氟化苯可获得氟化氧烷同分异构体。2025年2月20日出版的《自然-化学》发表了这项成果。
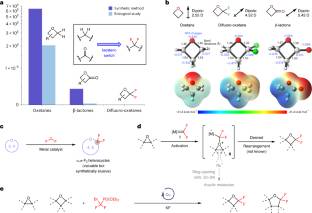
在这里,研究小组描述了一种铜催化的方法,通过与原位生成的二氟烃类反应来诱导碳原子插入氧杂环的骨架膨胀。根据计算得到的分子性质和静电电位图,α、α-二氟-氧辛烷产品是氧辛烷、β-内酯和羰基药效团的潜在替代品。这种方法的效用是突出的合成各种药物样分子和生物活性化合物的氟化同分异构体。实验和计算研究揭示了铜催化剂在促进反应开环和环化过程中的机理和独特作用。
研究人员表示,杂环构建块的骨架编辑提供了一种有吸引力的方法,通过多样化分子支架来扩大可访问的化学空间,用于药物发现。尽管最近在这一领域蓬勃发展,但由于应变诱导的环裂解和除氟的挑战,直接将氟引入小环杂环骨架的催化策略仍然很少。
附:英文原文
Title: Catalytic difluorocarbene insertion enables access to fluorinated oxetane isosteres
Author: Tan, Tong-De, Zhou, Fang, Quirion, Kevin P., Wang, Yu-Qi, Ng, Daniel Zhi Wei, Luo, Xiaohua, Chan, Eric Chun Yong, Liu, Peng, Koh, Ming Joo
Issue&Volume: 2025-02-20
Abstract: Skeletal editing of heterocyclic building blocks offers an appealing way to expand the accessible chemical space by diversifying molecular scaffolds for drug discovery. Despite the recent boom in this area, catalytic strategies that directly introduce fluorine into the backbone of small-ring heterocycles remain rare owing to the challenges of strain-induced ring cleavage and defluorination. Here we describe a copper-catalysed approach for skeletal expansion of oxygen heterocycles by reaction with a difluorocarbene species generated in situ to induce carbon atom insertion. The α,α-difluoro-oxetane products are potential surrogates of oxetane, β-lactone and carbonyl pharmacophores on the basis of their computed molecular properties and electrostatic potential maps. The utility of this approach is highlighted by synthesis of various drug-like molecules and fluorinated isosteres of biologically active compounds. Experimental and computational investigations provide insight into the mechanism and the unique role of the copper catalyst in promoting both ring-opening and cyclization steps of the reaction.
DOI: 10.1038/s41557-024-01730-7
Source: https://www.nature.com/articles/s41557-024-01730-7
Nature Chemistry:《自然—化学》,创刊于2009年。隶属于施普林格·自然出版集团,最新IF:24.274
官方网址:https://www.nature.com/nchem/
投稿链接:https://mts-nchem.nature.com/cgi-bin/main.plex
